Thrombus microstructure critically influences treatment success for conditions like stroke and deep vein thrombosis, yet current diagnostic methods struggle to fully capture these vital details. Hamed Ghodsi, Sara Cardona, Behrooz Fereidoonnezhad, and Sophinese Iskander-Rizk, all from Delft University of Technology, address this challenge with a new computational framework that generates realistic, virtual thrombi and simulates how they respond to photoacoustic imaging. Their work overcomes limitations in existing simulation platforms by accurately modelling microstructural features within larger, centimetre-scale phantoms at a reasonable computational cost. This innovative approach links the fine details of thrombus structure to the resulting imaging signals, offering a powerful tool for understanding disease progression and developing improved diagnostic techniques, and the team successfully demonstrates how thrombus microstructure significantly affects photoacoustic spectral responses.
Patient-Specific Clot Simulation via Photoacoustics
This research details a new approach to simulating thrombectomy procedures, the removal of blood clots, by combining photoacoustic imaging with detailed computer modeling. The goal is to create a virtual replica of a patient’s clot and surrounding blood vessels, allowing clinicians to plan personalized treatments and potentially improve clot removal effectiveness. Current thrombectomy planning relies on standard imaging techniques that lack detailed information about the internal structure of the clot, significantly impacting how it behaves during treatment. Researchers propose a workflow that integrates photoacoustic imaging with microstructure-based modeling, generating realistic three-dimensional models of the clot’s internal structure based on imaging data and known biological principles. This allows for simulation of clot mechanics and how it interacts with a thrombectomy device, ultimately promising improved treatment planning, optimized device designs, and a better understanding of clot mechanics.
Realistic Thrombus Generation via Recursive Framework
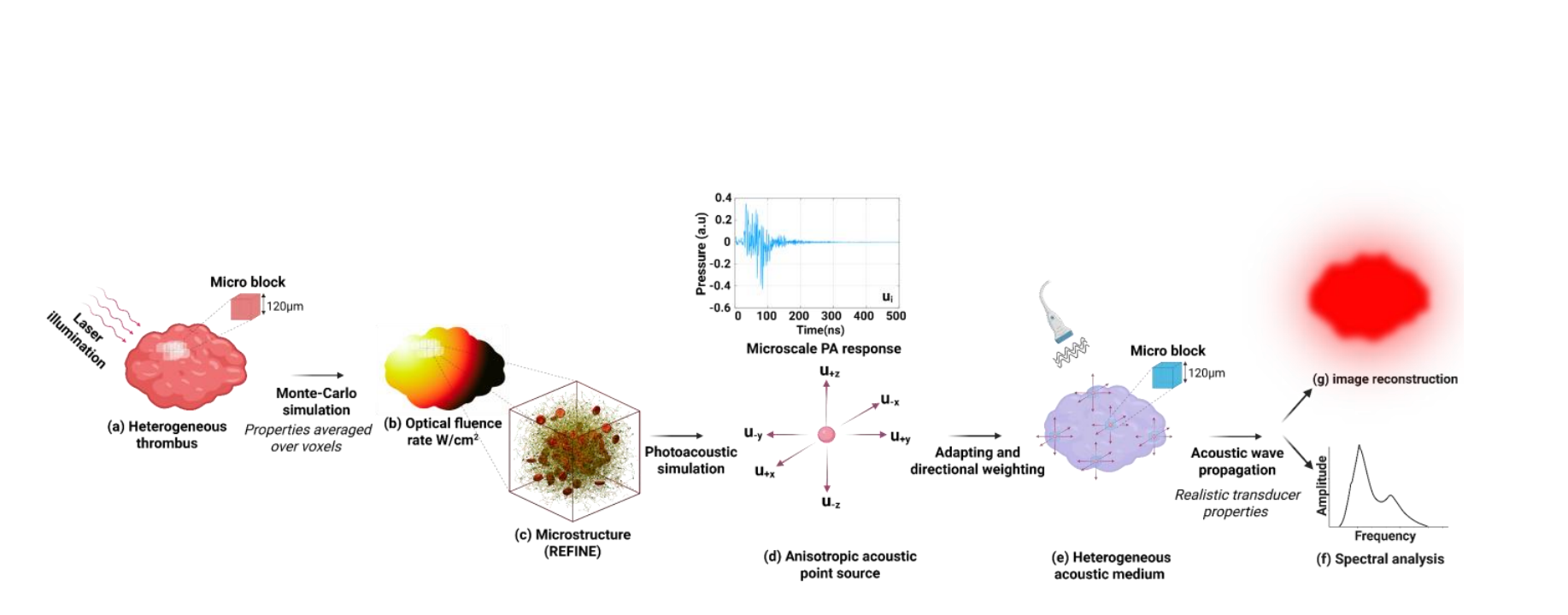
Scientists have developed REFINE, a new computational framework for generating realistic, computer-based models of blood clots, accurately replicating key internal features. Unlike existing methods, REFINE enables controlled and recursive optimization of clot structure, allowing for accurate photoacoustic modeling and potentially extending to biomechanical analyses. The team engineered this framework to create digital clots with defined compositions and porosities, overcoming limitations in sample availability and the challenges of experimentally characterizing these complex structures. Researchers embedded these digitally created clots within a multiscale photoacoustic simulation platform, bridging microscale acoustic modeling with macroscale clot geometries, effectively linking microstructural features to macroscale imaging outcomes and validating the approach against previous empirical studies.
Realistic Thrombus Models via Recursive Optimization
Scientists have developed REFINE, a new computational framework for generating realistic, computer-based models of blood clots, accurately replicating their complex internal structure. REFINE generates unique clot structures by employing a topology-driven recursive optimization process, allowing precise control over key features like fibrin fiber length, connectivity, and the distribution of red blood cells and platelets. The team successfully integrated these in silico clots into a multiscale photoacoustic simulation platform, bridging microscale acoustic modeling with macroscale clot geometries. This platform enables efficient and realistic simulation of photoacoustic signal responses, effectively linking microstructural features to macroscale imaging outcomes, and aligning with findings from previous empirical studies.
Virtual Thrombi and Photoacoustic Signal Prediction
This work presents REFINE, a new computational framework that generates realistic, virtual clots and simulates their photoacoustic signal responses. By combining biologically accurate clot generation with multiscale photoacoustic modeling, researchers demonstrate a clear link between clot microstructure and the resulting imaging signals. REFINE enables the creation of large datasets of virtual clots with customizable structure and varying compositions, facilitating the exploration of microstructure-related spectral biomarkers, and promising to advance diagnostic imaging strategies for thrombosis-related diseases, potentially paving the way for personalized treatment approaches guided by real-time clot characterization during intravascular interventions.
🗞 A Multiscale Framework for In Silico Thrombus Generation and Photoacoustic Simulations
🧠 ArXiv: https://arxiv.org/abs/2511.01642