The conventional understanding of molecular structure simplifies n-alk-1-ynes to a completely flat arrangement, but new research challenges this view. Ioan Bâldea, and colleagues demonstrate the existence of two distinct, nearly identical forms at the alkyne connection point, a planar structure and a skewed arrangement. The team’s comprehensive chemical analysis reveals a surprisingly high energy barrier to rotation, stemming from a unique combination of steric relief and electronic stabilisation, creating a ‘trigger finger’ effect that enforces a balance between these forms. This discovery fundamentally alters our understanding of alkyne behaviour, offering a pathway to accurately interpret spectroscopic data and achieve precise conformational control, which is particularly important for advancements in molecular electronics.
Skewed Alkynes Enable Molecular Switching
This research presents a detailed investigation into the structure of alkynes, revealing a surprising level of complexity at the molecular level. Contrary to the common assumption of a rigidly planar terminal carbon-carbon triple bond, scientists have demonstrated the existence of two near-isoenergetic conformations, planar and skewed, existing in equilibrium. This discovery challenges conventional understanding and opens new possibilities for controlling molecular behavior. The team’s findings demonstrate that the skewed conformation is kinetically stable, meaning it persists for measurable periods, creating a switchable element at the molecular interface.
The research highlights a surprisingly low energy barrier to rotation around the terminal carbon-carbon bond, significantly lower than that observed along the alkyl chain. This unique kinetic profile, likened to a “trigger finger,” allows for intentional enrichment of the skewed conformation during synthesis, providing a means to control the electronic properties of molecular junctions. This ability to manipulate conformation has significant implications for the field of molecular electronics, offering a pathway to design and optimize devices with tailored characteristics. The team’s analysis revealed that the low rotational barrier stems from a unique interplay of steric relief and electronic stabilization.
The absence of hydrogen atoms on one of the carbon atoms involved in the terminal bond reduces steric hindrance, while electronic effects contribute to the stability of the skewed conformation. This combination results in a conformational landscape distinct from that of traditional alkyl chains. The research provides a comprehensive understanding of the factors governing alkyne conformation, offering a solid foundation for future investigations and applications.
Alkyne Rotamers Persist at Room Temperature
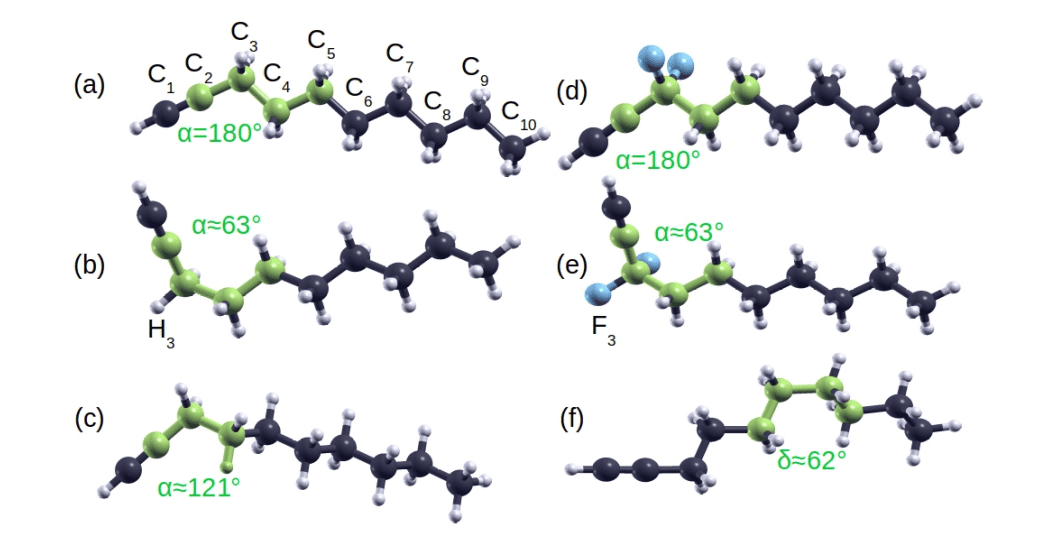
This work presents a groundbreaking re-evaluation of alkyne conformation, revealing a surprising level of structural complexity at the acetylenic terminus. Contrary to the conventional assumption of a purely planar structure, scientists achieved definitive evidence of two near-isoenergetic rotamers, planar and skewed, existing in equilibrium. Detailed quantum chemical analysis consistently demonstrated that these conformers are separated by a symmetric rotational energy barrier of approximately 150 meV, establishing their kinetic persistence at room temperature. This barrier height remains remarkably consistent across varying chain lengths, indicating an intrinsic molecular property rather than a chain-length dependent effect.
The research demonstrates that the energetic difference between the planar and skewed conformers is minimal, consistently below 0. 2 kcal/mol across a range of alkyne sizes. This finding was confirmed by multiple high-level computational methods and validated by analysis using a polarizable continuum model, which confirmed the robustness of the conformational landscape in diverse solvents. Measurements of bond metrics for both conformers provided a detailed structural basis for understanding this unique behavior. The team discovered a significant difference in the rotational profile between internal rotation and the terminal rotation.
The terminal rotation, involving a bond between sp-hybridized carbon atoms, exhibits a suppressed steric penalty due to the absence of hydrogen atoms, providing substantial steric relief. This contrasts sharply with the traditional gauche penalty observed in rotations involving sp3-hybridized carbon atoms. The discovery of this unique interplay between steric relief and electronic stabilization fundamentally alters our understanding of alkyne conformation. This research establishes a conformational profile driven by internal molecular properties, opening new avenues for controlling molecular structure and reactivity through subtle conformational changes.
Alkynes Exhibit Dual Rotameric Conformation
This research redefines the conventional understanding of molecular conformation in alkyne molecules, demonstrating the existence of two near-isoenergetic rotamers at the acetylenic terminus, planar and skewed, rather than a single, fully planar structure. The team discovered a unique rotational energy barrier arising from steric relief and electronic stabilization, resulting in a distinctive “trigger finger” rotation, contrasting with the behavior of the alkyl chain. This structural degeneracy necessitates ensemble averaging when interpreting spectroscopic data, as the two conformers coexist, yet the slow interconversion allows for kinetic trapping and intentional enrichment during synthesis. The findings have significant implications for molecular electronics, particularly in the design and interpretation of experiments involving alkyne-based molecular junctions.
By revealing conformational control within these molecules, the work provides a blueprint for accurate data interpretation and opens avenues for tailoring molecular properties for specific electronic applications. Future research could focus on exploring the influence of different substrates and external stimuli on these rotamers, potentially leading to the development of novel molecular devices with enhanced performance and functionality. The research establishes a new paradigm for understanding alkyne conformation, paving the way for the design of more sophisticated molecular materials and devices. The ability to control and manipulate molecular conformation opens up exciting possibilities for a wide range of applications, from organic electronics to materials science.
👉 More information
🗞 Unlocking n-alk-1-ynes Conformers: Quantum “Trigger Finger” versus “Stiff Joint” Conformations
🧠 ArXiv: https://arxiv.org/abs/2511.02470